Compound Semiconductor Optoelectronics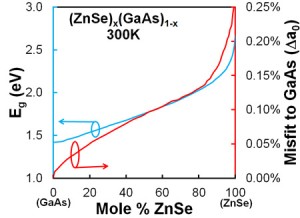 A significant portion of our research is focused on photonic devices using compound semiconductors. We have worked on a variety of devices (solar cells, laser diodes, LEDs, photodetectors, etc.) using a variety of materials (GaAs, InP, GaP, GaN, etc.). Our latest research includes obtaining high purity GaP by gettering in Al-Ga melts, obtaining large Schottky barrier heights on p-GaAs for MIS-based solar cells, and developing a world-record ZnO/InP heterojunction solar cell. We will continue research in these areas while also exploring additional topics such as studying the ZnSe-GaAs material system for true green LEDs, and developing Al-rich AlGaInP films by liquid phase epitaxy for lattice-matched, wide band gap solar cells and LEDs. Hybrid Photovoltaic-Photothermal Systems Using Selective Absorbers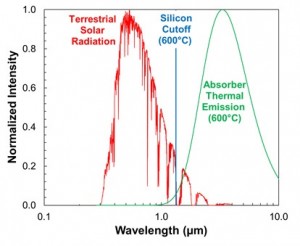 In most traditional photovoltaic systems, heat is concerned to be a hindrance to achieving maximum energy conversion efficiency. Typically, solar cells are cooled by mounting onto a passively or actively-cooled conductive plate, resulting in all of the heat energy being wasted in the system. By evaluating the use of semiconductors for selective absorbers, we are investigating novel systems that could integrate both a high-temperature photovoltaic cell with a photothermal device. The key to success is in careful design of a high temperature solar cell coupled to a selective absorber for capture and conversion of the heat energy. This design offers an alternative to high efficiency, concentrated photovoltaics (CPV) where a very expensive multijunction solar cell is essentially replaced with a single junction solar cell and selective absorber. Water Splitting Using Earth-Abundant Aluminum Alloys Over the past decade, there has been a sharp intensification of search for a new source of energy, and hydrogen power has been one of the alternative solutions. To introduce hydrogen in a technological cycle, it is necessary to solve three most essential problems: receiving, storage and transportation, and hydrogen utilization. At present, the most imminent problem lies in the storage and transportation part. One of the possible perspective solutions to this problem is to store the hydrogen in a latent form and produce it directly at the location of consumption, i.e. combine the receiving stage and consumption stage in one system or one device. 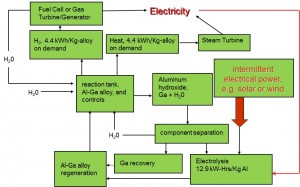 The aluminum-gallium alloy is capable of closing this three stage cycle. The aluminum-gallium alloy by itself does not contain hydrogen. However when water is introduced to the system, the alloy will react and hydrogen will be generated by splitting water. The oxygen component will combine with the aluminum component of the alloy and produce aluminum hydroxide which can be recycled back into aluminum. The gallium component is inert and is also recyclable. The benefit of this system involves ease of storage and transportation. Instead of storing and transporting hydrogen directly which can be dangerous and expensive, this system allows the user to transport solid alloys. The solid alloys are inert in air and will only generate hydrogen when water is introduced, thus allowing the user to generate hydrogen directly at the location of consumption. The overall process is economic as gallium are inert and can be reused and aluminum can be reproduced from aluminum hydroxide. See this work in action on our media page. |
CONTACT US
For General Inquiries, contact Prof. Woodall (jwoodall@ucdavis.edu).
LOCATION
Our lab is in Ghausi Hall, 1123
Mailing Address:
UC Davis – ECE Department
ATTN: Woodall Group
2064 Kemper Hall
One Shields Avenue
Davis, CA 95616